 Selasa, 30 April 2024 (14:56)
Selasa, 30 April 2024 (14:56)
 Music |
 Video |
 Movies |
 Chart |
 Show |
 |
The following redox reaction occurs in basic medium `NO (3)^(-)Zn(s)toZn^(2+)+NH (4)^(+)` when the (Doubtnut) View |
 |
For the redox reaction, Zn + NO (3)^(-) to Zn^(2+) + NH (4)^(+) In basic medium, coefficients of... (Doubtnut) View |
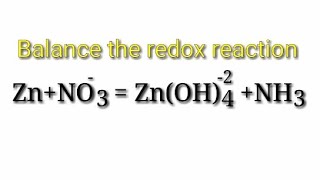 |
Balance the redox reaction by ion electron method or half reaction method. Zn+NO3-=Zn(OH)4-2+NH3. (My documentary) View |
 |
Using Half-Reactions to Balance Equations in Basic Solution (Professor Johnston) View |
 |
Ag+Zn^2+=Ag2O+Zn balance the redox reaction by ion electron method @mydocumentary838 (My documentary) View |
 |
Balancing redox reaction in a basic environment (Eric Victor) View |
 |
Balance IO3- (Aq) + I- (Aq) → I2 (S) - Redox Reactions in Acidic media (Transcended Institute) View |
 |
17.6d | Balance the equation in an basic solution: Al(s) + CrO42−(aq) → Al(OH)3(s) + Cr(OH)4−(aq) (The Glaser Tutoring Company) View |
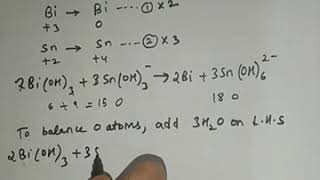 |
11th STD Redox Part 07 (Chemistry with Kalpana Jajoo) View |
 |
Balance the Redox Reaction Cu + HNO3 → Cu(NO3)2 + NO + H2O (Wayne Breslyn) View |